 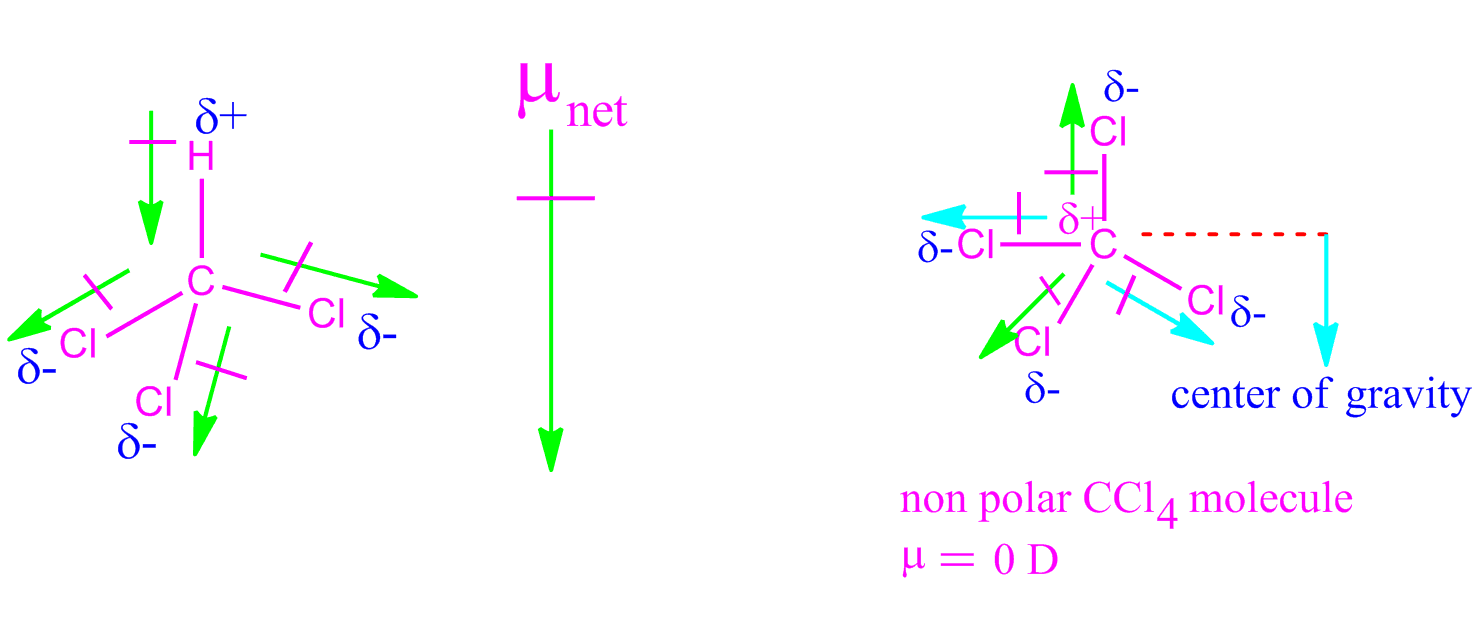
We just discussed examples where the dipole moments were enforced by one another according to their vector sum. The dipole moment of the lone pair is reinforced by the vector sum of the three N-H bonds: Similarly, ammonia has a greater dipole moment (1.47 D) than each N-H bond itself (1.3 D). Therefore, 1.85 D is the vector sum of the two dipoles of 1.5 D at 104.5 o plus the reinforcing effect of the lone pairs. It is very helpful to learn the hybridization and VSEPR theories at his point which suggest that the hydrogens and the lone pairs on the oxygen are in sp 3 orbitals at ~109.5 o. This can be proved by the greater value of the water dipole moment (1.85 D) compared to the dipole moment of the individual O-H bonds (1.5 D). Although the resulting dipole vectors are not linearly aligned at 0 o, they all point in the same direction, and when combining them, we see that all the dipoles reinforce each other. 
The lone pairs, on the other hand, point the dipole moment towards them. Oxygen pulls the bonding electrons shared with the hydrogens since it is more electronegative. It is important to mention that lone pairs of electrons also affect the magnitude and direction of the molecular dipole.įor example, the water molecule has two polar bonds and one lone pair of electrons. Now, if there are multiple polar bonds, the molecular dipole moment is determined by the vector sum of those dipole moments.įor example, if we replace one more hydrogen atom in dichloromethane (CH 2Cl 2 shown above), we need to find the vector sum of the two C-Cl dipoles to determine the overall dipole of the molecule: Remember, for molecules with one polar bond, the molecular dipole is determined simply based on the dipole moment of that bond: The dipole moment of a molecule and its overall polarity depends on the magnitude and direction of individual polar bonds and their dipole moments. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
